The Extremely-Small Chamber (ULC) Series has the ability to freeze product or service from ambient temperatures to -eighty°C, but it doesn't enable for specific cooling profiles such as fall and hold or controlled temperature improvements/minute.
Looking ahead to 2025, we are able to hope to check out even higher integration of automation in cleanroom sterilization.
We also give demonstration models to check, determine operational procedures about, and program workflows. Make contact with us to ascertain The easiest way to guidance your challenge needs.
Productive staff schooling for cleanroom sterilization goes over and above procedural expertise; it will have to instill a deep idea of contamination pitfalls and foster a lifestyle of top quality and compliance.
For centrifugal air samplers, many before reports confirmed that the samples shown a selectivity for more substantial particles. Using such a sampler might have resulted in better airborne counts as opposed to other types of air samplers because of that inherent selectivity.
What forms of life science products and drug merchandise are saved in FARRAR’s engineered wander-in rooms?
Designed with Repeated entry storage, scientific demo kitting, and logistics prep system requires in your mind
The initial space in which the raw and packaging components are exposed to the room environment is in the incoming sampling room. Right here containers needs to be opened to consider samples with the Uncooked and packaging resources. The requirements for this region have to be the same as within the production space which is often ISO 8 Course one hundred,000. The changeover point among a warehouse along with the manufacturing region will be the dispensary or weigh room.
Constant schooling has become increasingly vital as cleanroom engineering evolves. Several corporations are applying frequent refresher courses and competency assessments in order that cleanroom in pharmaceutical industry staff expertise remain up-to-day.
This method delivers automated defrosting capabilities and quick repairs devoid of getting freezers out of provider. An ordinary cold wall ULT freezer relies on organic convection to chill its chamber, a slower and less uniform approach.
Clean rooms are divided into groups ISO one by means of ISO nine in accordance with the variety of allowable particles for every cubic meter. These environments are vital for your manufacture of all sterile merchandise for example injectables, vaccines, and biologics.
Good Force. Sophisticated mechanical programs constantly drive air into your room to blow out airborne contaminants. Pressure, similar to temperature, is recorded each day and will be examine from outside the room.
The main aim for just a design group is to lessen or do away with the potential of more info contamination within a cleanroom. 1 critical to defense is acquiring airlocks involving the process spot plus the adjacent space. This can help guard the product when getting transferred from room to room.
Chance Evaluation Analysis —Evaluation with the identification of contamination potentials in managed environments that set up priorities concerning severity and frequency and that can develop procedures and treatments that should eradicate, lower, limit, or mitigate their probable for microbial contamination with the merchandise/container/closure program.
 Alicia Silverstone Then & Now!
Alicia Silverstone Then & Now!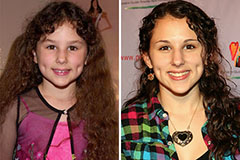 Hallie Eisenberg Then & Now!
Hallie Eisenberg Then & Now! Kelly McGillis Then & Now!
Kelly McGillis Then & Now! Ross Bagley Then & Now!
Ross Bagley Then & Now! Soleil Moon Frye Then & Now!
Soleil Moon Frye Then & Now!